Published by: VITA 1600 Wilson Boulevard, Suite 500 Arlington, Virginia 22209 USA Tel: 703/276-1800 * Fax: 703/243-1865 Internet: pr-info@vita.org
PREFACE
This paper is one of a series published by Volunteers in Technical Assistance to provide an introduction to specific state-of-the-art technologies of interest to people in developing countries. The papers are intended to be used as guidelines to help people choose technologies that are suitable to their situations. They are not intended to provide construction or implementation details. People are urged to contact VITA or a similar organization for further information and technical assistance if they find that a particular technology seems to meet their needs.
The papers in the series were written, reviewed, and illustrated almost entirely by VITA Volunteer technical experts on a purely voluntary basis. Some 500 volunteers were involved in the production of the first 100 titles issued, contributing approximately 5,000 hours of their time. VITA staff included Maria Giannuzzi as editor, Suzanne Brooks handling typesetting and layout, and Margaret Crouch as project manager.
The author of this paper, VITA Volunteer Horace McCracken, is the president of the McCracken Solar Company in Alturas, California. The co-author, VITA Volunteer Joel Gordes, is currently the solar design analyst for the State of Connecticut's Solar Mortgage Subsidy Program. The reviewers are also VITA volunteers. Daniel Dunham has done consulting in solar and alternative sources of energy for VITA and AID. He has lived and worked in India, Pakistan, and Morocco. Mr. Dunham has also prepared a state-of-the-art survey on solar stills for AID. Jacques Le Normand is Assistant Director at the Brace Research Institute, Quebec, Canada, which does research in renewable energy. He has supervised work with solar collectors and has written several publications on solar and wind energy, and conservation. Darrell G. Phippen is a mechanical engineer and development specialist who works with Food for the Hungry in Scottsdale, Arizona.
VITA is a private, nonprofit organization that supports people working on technical problems in developing countries. VITA offers information and assistance aimed at helping individuals and groups to select and implement technologies appropriate to their situations. VITA maintains an international Inquiry Service, a specialized documentation center, and a computerized roster of volunteer technical consultants; manages long-term field projects; and publishes a variety of technical manuals and papers. For more information about VITA services in general, or the technology presented in this paper, contact VITA at 1815 North Lynn Street, Suite 200, Arlington, Virginia 22209 USA.
I. INTRODUCTION
Ninety-seven percent of the earth's water mass lies in its oceans. Of the remaining 3 percent, 5/6 is brackish, leaving a mere .5 percent as fresh water. As a result, many people do not have access to adequate and inexpensive supplies of potable water. This leads to population concentration around existing water supplies, marginal health conditions, and a generally low standard of living.
Solar distillation uses the heat of the sun directly in a simple piece of equipment to purify water. The equipment, commonly called a solar still, consists primarily of a shallow basin with a transparent glass cover. The sun heats the water in the basin, causing evaporation. Moisture rises, condenses on the cover and runs down into a collection trough, leaving behind the salts, minerals, and most other impurities, including germs.
Although it can be rather expensive to build a solar still that is both effective and long-lasting, it can produce purified water at a reasonable cost if it is built, operated, and maintained properly.
This paper focuses mainly on small-scale basin-type solar stills as suppliers of potable water for families and other small users. Of all the solar still designs developed thus far, the basin-type continues to be the most economical.
HISTORY OF SOLAR DISTILLATION
Distillation has long been considered a way of making salt water drinkable and purifying water in remote locations. As early as the fourth century B.C., Aristotle described a method to evaporate impure water and then condense it for potable use.
P.I. Cooper, in his efforts to document the development and use of solar stills, reports that Arabian alchemists were the earliest known people to use solar distillation to produce potable water in the sixteenth century. But the first documented reference for a device was made in 1742 by Nicolo Ghezzi of Italy, although it is not known whether he went beyond the conceptual stage and actually built it.
The first modern solar still was built in Las Salinas, Chile, in 1872, by Charles Wilson. It consisted of 64 water basins (a total of 4,459 square meters) made of blackened wood with sloping glass covers. This installation was used to supply water (20,000 liters per day) to animals working mining operations. After this area was opened to the outside by railroad, the installation was allowed to deteriorate but was still in operation as late as 1912--40 years after its initial construction. This design has formed the basis for the majority of stills built since that time.
During the 1950s, interest in solar distillation was revived, and in virtually all cases, the objective was to develop large centralized distillation plants. In California, the goal was to develop plants capable of producing 1 million gallons, or 3,775 cubic meters of water per day. However, after about 10 years, researchers around the world concluded that large solar distillation plants were much too expensive to compete with fuel-fired ones. So research shifted to smaller solar distillation plants.
In the 1960s and 1970s, 38 plants were built in 14 countries, with capacities ranging from a few hundred to around 30,000 liters of water per day. Of these, about one third have since been dismantled or abandoned due to materials failures. None in this size range are reported to have been built in the last 7 years.
Despite the growing discouragement over community-size plants, McCracken Solar Company in California continued its efforts to market solar stills for residential use. Worldwide interest in small residential-units is growing, and now that the price of oil is ten times what it was in the 1960s, interest in the larger units may be revived.
Although solar distillation at present cannot compete with oil-fired desalination in large central plants, it will surely become a viable technology within the next 100 years, when oil supplies will have approached exhaustion. When that day arrives, the primary question will be, "Which method of solar distillation is best?" Meanwhile, almost anyone hauling drinking water any distance would be economically better off using a solar still.
NEEDS SERVED BY SOLAR DISTILLATION
Solar distillation could benefit developing countries in several ways:
- Solar distillation can be a cost-effective means of providing clean water for drinking, cooking, washing, and bathing--four basic human needs.
- It can improve health standards by removing impurities from questionable water supplies.
- It can help extend the usage of existing fresh water in locations where the quality or quantity of supply is deteriorating. Where sea water is available, it can reduce a developing country's dependence on rainfall.
- Solar stills, operating on sea or brackish water, can ensure supplies of water during a time of drought.
- Solar distillation generally uses less energy to purify water than other methods.
- It can foster cottage industries, animal husbandry, or hydroponics for food production in areas where such activities are now limited by inadequate supplies of pure water. Fishing could become important on desert seacoasts where no drinking water is available for fishermen.
- Solar distillation will permit settlement in sparsely-populated locations, thus relieving population pressures in urban areas.
APPLICATIONS
The energy from the sun used to distill water is free. But the cost of building a still makes the cost of the distilled water rather high, at least for large-scale uses such as agriculture and flushing away wastes in industry and homes. Consequently, the solar still is used principally to purify water for drinking and for some business, industry, laboratory, and green-house applications. It also appears able to purify polluted water.
Solar Distilled Water for Irrigation
For field agriculture, the solar still is not very promising. It takes about one meter depth of irrigation water per year to produce crops in dry climates, whereas the solar still can evaporate about two meters' depth. Thus, one square meter of solar still would irrigate two square meters of land. Unquestionably, the cost of building the still would make water more valuable than the crops being produced. This may not be true, however, for agriculture in controlled environments, i.e., greenhouses. A well-designed hydroponically-operated greenhouse should be able to produce 8 to 10 times as much food, per unit volume of water consumed, as field crops.
Recovery of Salt from a Solar Still
Since salt is a very cheap industrial material, and a solar still cannot produce anymore than an open pond, combining the recovery of salt with the distilling of water is not attractive economically. Where a family is using a solar still to provide water valued at $1 per day, the amount of salt they need might cost them half a cent.
Recovery of Potable Water from Sewage
Although it seems possible that potable water can be recovered from sewage, if contaminants such as odorous gases are present in sewage water fed to the still, some portion of those gases will evaporate and condense with the distilled water. In all probability they could be filtered out with activated carbon, but to date, however, no one has had any experience with this.
Alcohol Production
If the "contaminant" is alcohol, it can be separated from the water. But it would take two or three passes through the still to attain a high enough concentration of alcohol to be used as a fuel. Considering the current availability of fossil fuels, producing alcohol in this way is not yet economical. However, when fossil fuel supplies run low and the price rises, solar distillation could play a significant role.
Recovery of Distilled Water From Polluted Water Bodies
Whether or not solar distillation can actually purify polluted water is not yet known. Laboratory tests have shown, however, that a solar still can eliminate bacteria. If after additional research, a quantity of clean water can be recovered from polluted water, this capability may become economically more important than the purification of sea water. It may also be used to remove toxic substances such as pesticides.
Preliminary laboratory tests show that a modified version of the still--now commercially available--can do a very good job of removing such substances from feed water. Trichloroethylene (TCE), for example, has been removed by a factor of 5,000 to 1; ethylene dibromide (EDB) by 100 to 1; nitrates by 50 to 1; and others within those ranges. Of course, more work must be done to quantify these numbers, not to mention the unending list of chemicals that need to be tested.
Elimination of Algae. While algae will grow in some deep basin stills where the water temperature seldom gets very high, in the shallow basin still it is usually killed by the high temperature.
II. OPERATING PRINCIPLES
GENERAL THEORY OF SOLAR DISTILLATION
Distillation operates by the escape of moving molecules from the water surface into the gases above it. Sensible heat--the kind you can measure with a thermometer--is caused by the movement of molecules, zig-zagging about constantly, except that they are not all moving at the same speed. Add energy and they move faster, and the fastest-moving ones may escape the surface to become vapor.
It takes a lot of energy for water to vaporize. While a certain amount of energy is needed to raise the temperature of a kilogram of water from 0 [degrees] to 100 [degrees] Celsius (C), it takes five and one-half times that much to change it from water at 100 [degrees] C to water vapor at 100 [degrees] C. Practically all this energy, however, is given back when the water vapor condenses.
The salts and minerals do not evaporate along with the water. Ordinary table salt, for example, does not turn into vapor until it gets over 1400 [degrees] C, so it remains in the brine when the water evaporates. This is the way we get fresh water in the clouds from the oceans, by solar distillation. All the fresh water on earth has been solar distilled.
It is not necessary for the water to actually boil to bring about distillation. Steaming it away gently does the same job as boiling, except that in the solar still, it will usually turn out even more pure, because during boiling the breaking bubbles may contaminate the product water with tiny droplets of liquid water swept along with the vapor.
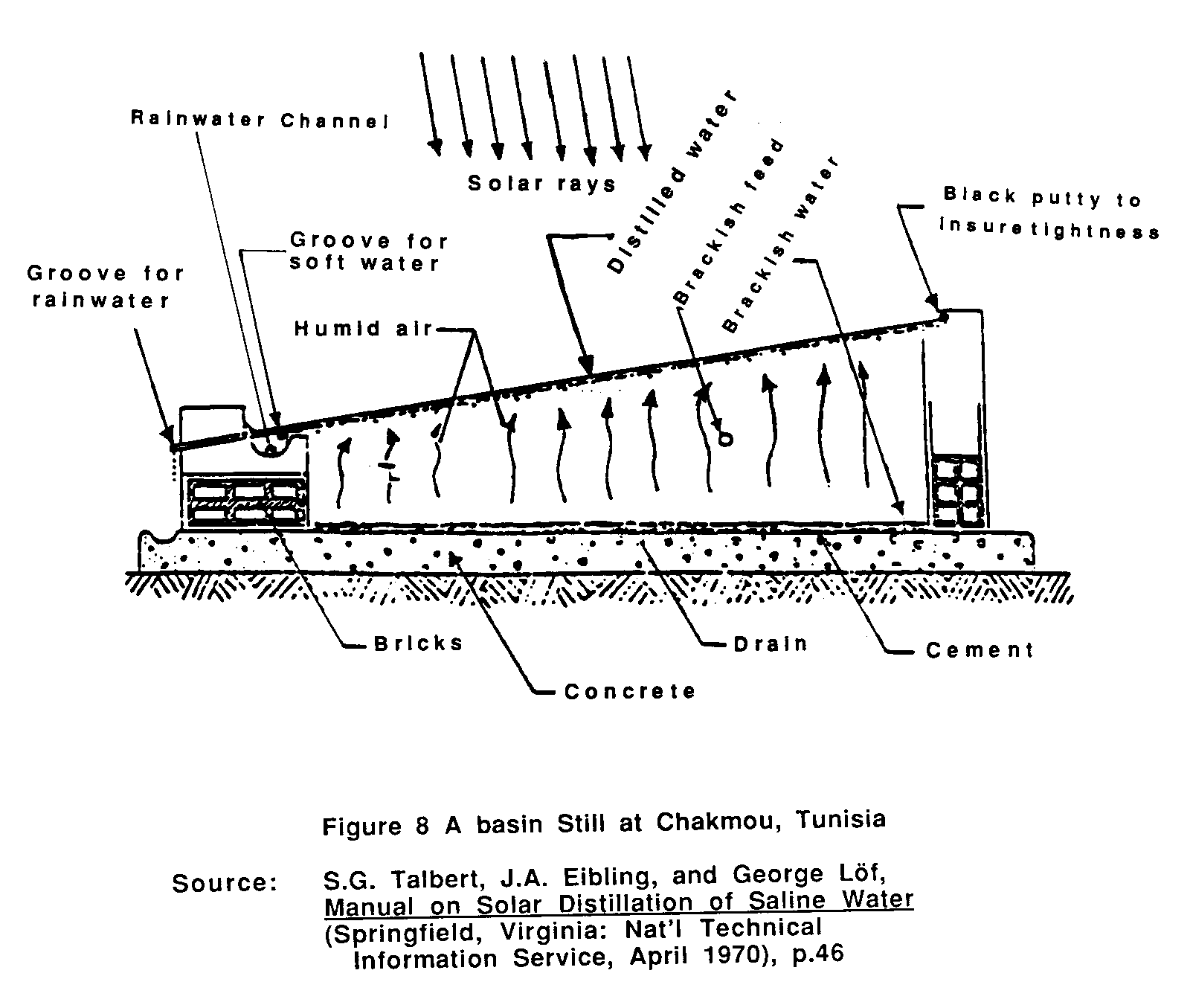
THE SOLAR DISTILLATION PROCESS
The solar distillation process is shown in Figure 1. Solar
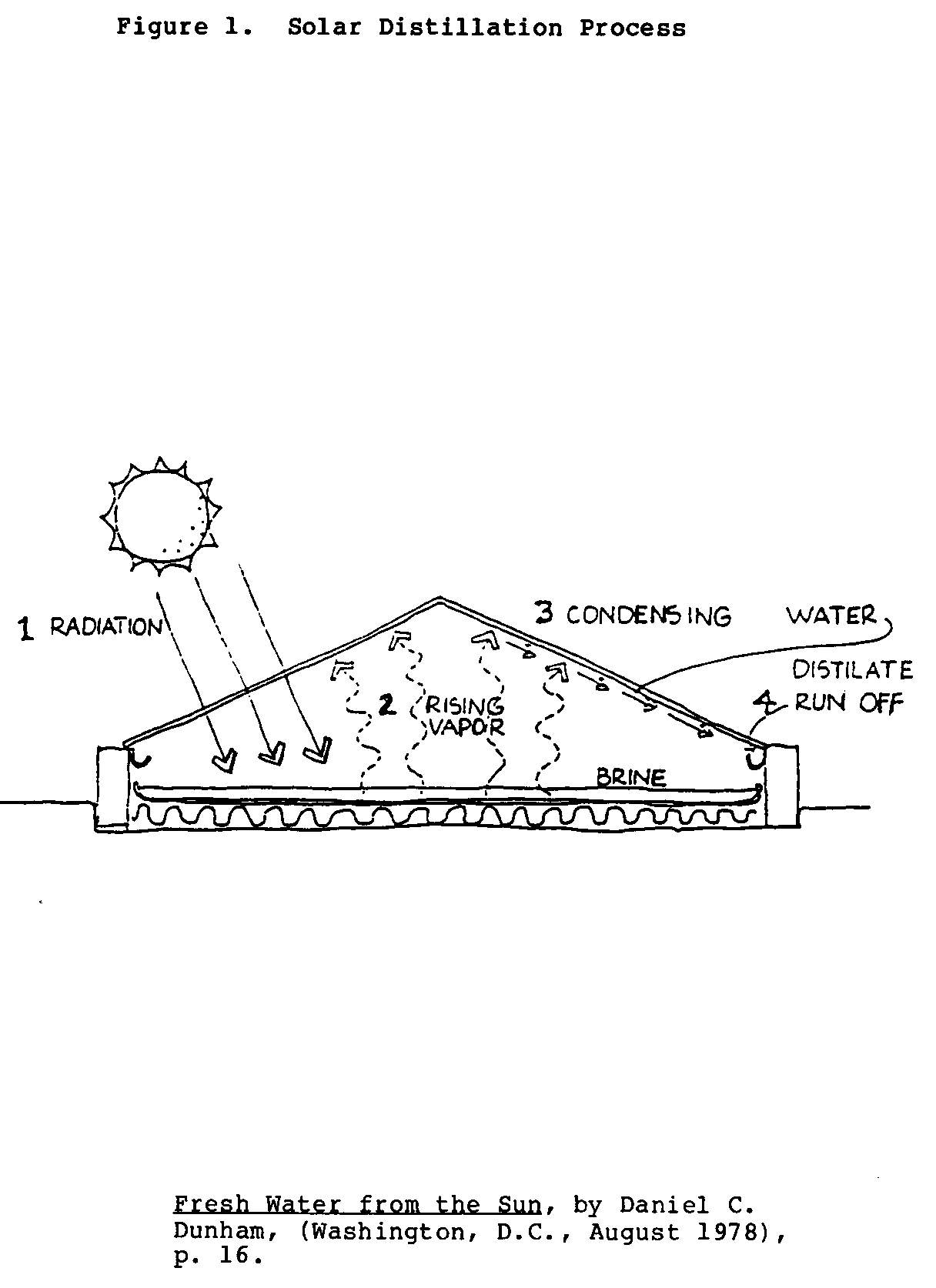
energy passing through a glass cover heats up the brine or sea water in a pan; this causes the water to vaporize. The vapor then rises and condenses on the underside of the cover and runs down into distillate troughs.
Fresh Water from the Sun, by Daniel C. Dunham, (Washington, D.C., August 1978), p. 16. A more technical description follows.:
1. The sun's energy in the form of short electromagnetic waves passes through a clear glazing surface such as glass. Upon striking a darkened surface, this light changes wavelength, becoming long waves of heat which is added to the water in a shallow basin below the glazing. As the water heats up, it begins to evaporate.
2. The warmed vapor rises to a cooler area. Almost all impurities are left behind in the basin.
3. The vapor condenses onto the underside of the cooler glazing and accumulates into water droplets or sheets of water.
4. The combination of gravity and the tilted glazing surface allows the water to run down the cover and into a collection trough, where it is channeled into storage.
In most units, less than half the calories of radiant energy falling on the still are used for the heat of vaporization necessary to produce the distilled water. A commercial stills are sold to date have had an efficiency range of 30 to 45 percent. (The maximum efficiency is just over 60 percent.) Efficiency is calculated in the following manner:
Energy required for the vaporization of the distillate that is recovered Efficiency = Energy in the sun's radiation that falls on the still.
Providing the costs don't rise significantly, an efficiency increase of a few percent is worth working for. Improvements are principally to be sought in materials and methods of construction.
III. SOLAR STILL DESIGN VARIATIONS
Although there are many designs for solar stills, and four general categories, (concentrating collector stills; multiple tray tilted stills; tilted wick solar stills; and basin stills) 95 percent of all functioning stills are of the basin type.
CONCENTRATING COLLECTOR STILL
A concentrating collector still, as shown in Figure 2, uses
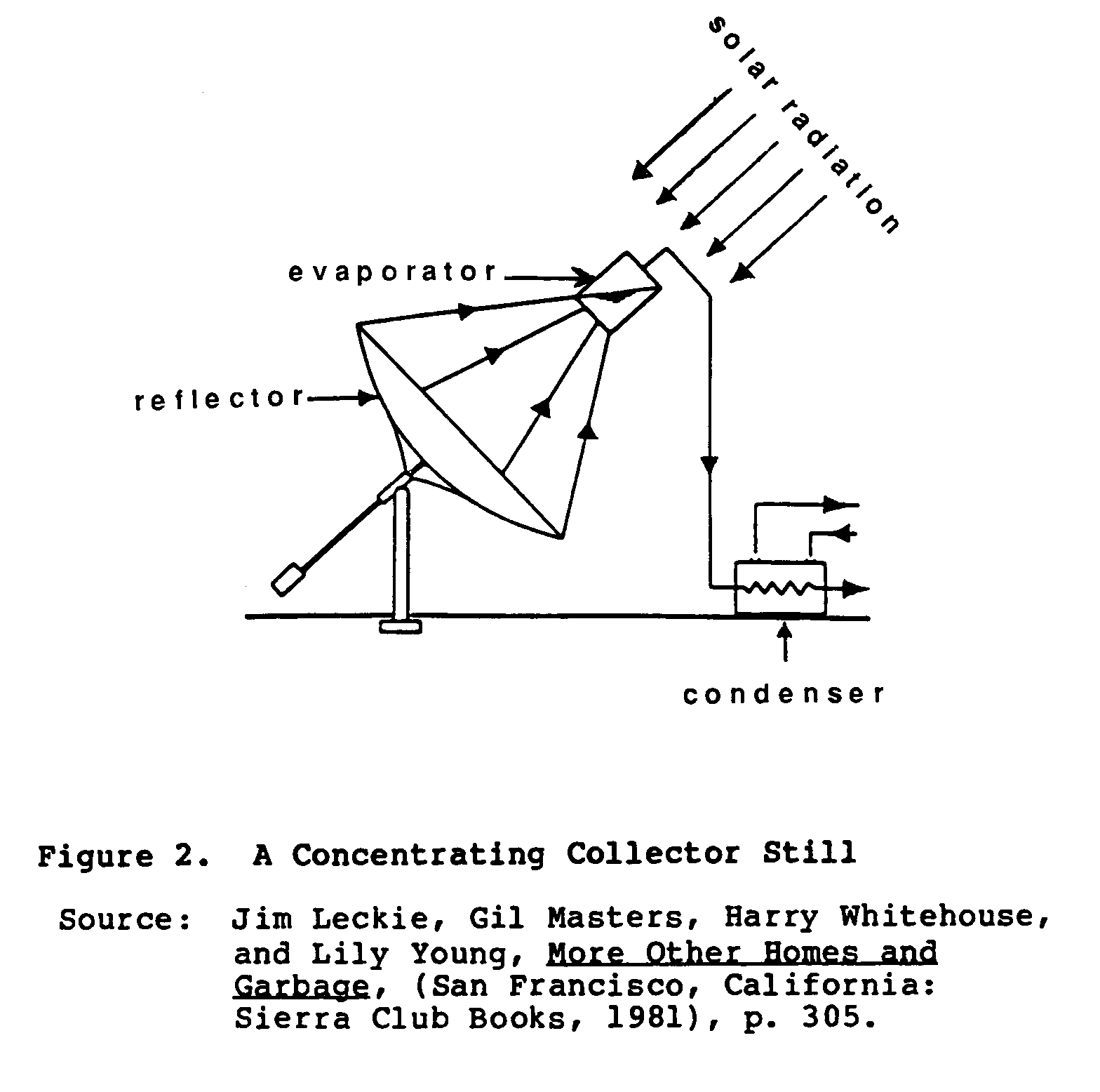
parabolic mirrors to focus sunlight onto an enclosed evaporation vessel. This concentrated sunlight provides extremely high temperatures which are used to evaporate the contaminated water. The vapor is transported to a separate chamber where it condenses, and is transported to storage. This type of still is capable of producing from .5 to .6 gallons per day per square foot of reflector area. This type of output far surpasses other types of stills on a per square foot basis. Despite this still's outstanding performance, it has many drawbacks; including the high cost of building and maintaining it, the need for strong, direct sunlight, and its fragile nature.
MULTIPLE TRAY TILTED STILL
A multiple tray tilted still (Figure 3), consists of a series of

shallow horizontal black trays enclosed in an insulated container with a transparent top glazing cover. The vapor condenses onto the cover and flows down to the collection channel for eventual storage.
This still can be used in higher latitudes because the whole unit can be tilted to allow the sun's rays to strike perpendicular to the glazing surface. The tilt feature, however, is less important at and near the equator where there is less change in the sun's position over the still. Even though efficiencies of up to 50 percent have been measured, the practicality of this design remains doubtful due to:
- the complicated nature of construction involving many components;
- increased cost for multiple trays and mounting requirements.
TILTED WICK SOLAR STILL
A tilted wick solar still draws upon the capillary action of fibers to distribute feed water over the entire surface of the wick in a thin layer. The water is then exposed to sunlight. (See Figure 4.)
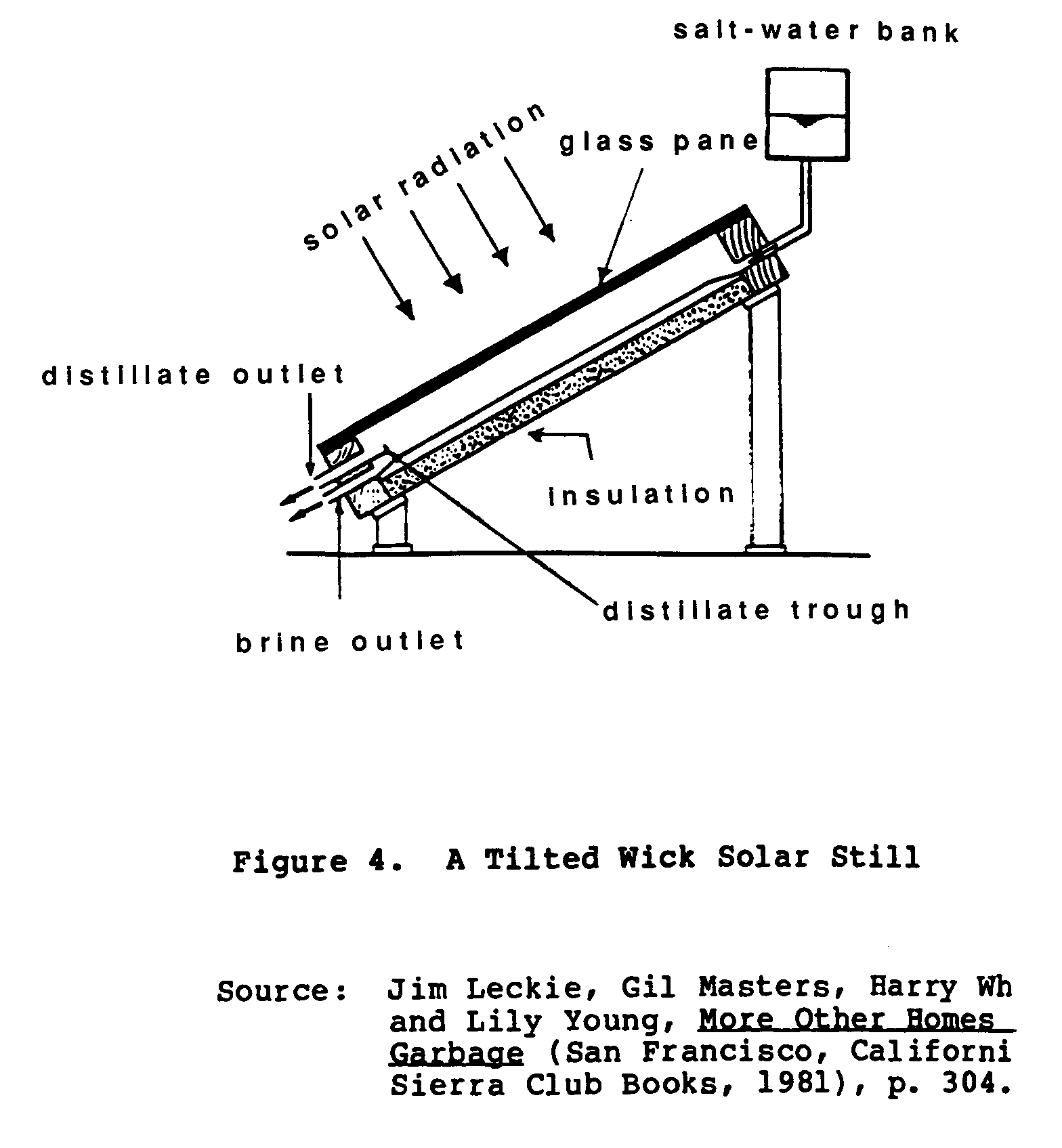
A tilted wick solar still allows a higher temperature to form on this thin layer than can be expected from a larger body of water. This system is as efficient as the tilted tray design, but its use in the field remains questionable because of:
- increased costs due to mounting requirements and essential insulation;
- the need to frequently clean the cloth wick of built-up sediments, highlighting the need for an operable glazing cover;
- the need to replace the black wick material on a regular basis due to sun bleaching and physical deterioration by ultra-violet radiation;
- uneven wetting of the wick which will result in dry spots, leading to reduced efficiency; and
- the unnecessary aspect of the tilt feature except where it is required higher latitudes.
BASIN STILL
A basin still (See Figure 5), is the most common type in use,
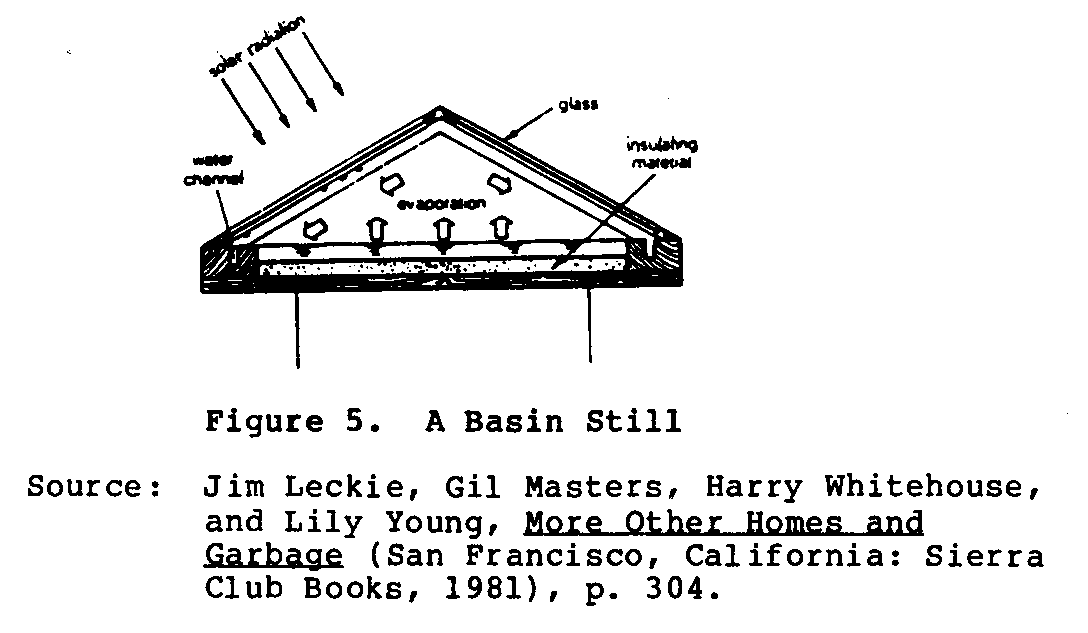
although not in current production.
While the basic design can take on many variations, the actual shape and concept have not changed substantially from the days of the Las Salinas, Chile stills built in 1872. The greatest changes have involved the use of new building materials, which may have the potential to lower overall costs while providing an acceptably long useful life and better performance.
All basin stills have four major components:
1. a basin; 2. a support structure; 3. a transparent glazing cover; and 4. a distillate trough (water channel).
In addition to these, ancillary components may include:
1. insulation (usually under the basin); 2. sealants; 3. piping and valves; 4. facilities for storage; 5. an external cover to protect the other components from the weather and to make the still esthetically pleasing; and 6. a reflector to concentrate sunlight.
Physical Dimensions of the Basin Still
The actual dimensions of basin stills vary greatly, depending on the availability of materials, water requirements, ownership patterns, and land location and availability.
If the only glazing available is one meter at its greatest dimension, the still's maximum inner width will be just under one meter. And the length of the still will be set according to what is needed to provide the amount of square meters to produce the required amount of water. Likewise, if an entire village were to own and use the still, the total installation would have to be quite large.
It is generally best to design an installation with many small modular units to supply the water. This allows:
- units to be added;
- manageable components to be handled by unskilled persons without expensive mechanical equipment;
- maintenance can be carried out on some units while others continue to operate.
Most community size stills 1/2 to 21/2 meters wide, with lengths ranging up to around 100 meters. Their lengths usually run along an eastwest axis to maximize the transmission of sunlight through the equatorialfacing sloped glass. Residential, appliance type units generally use glass about 0.65 to 0.9 meter wide with lengths ranging from two to three meters. A water depth of 1.5 to 2.5 cm is most common.
The usual argument for greater depths is that the stored heat can be used at night to enhance production when the air temperatures are lower. Unfortunately, no deep basin has ever attained the 43 percent efficiency typical of a still of minimum water depth. The results to date are clear: the shallower the depth the better. Of course, if the basin is too shallow, it will dry out and salts will be deposited, which is not good. Note that solar heat can evaporate about 0.5 cm of water on a clear day in summer. By setting the initial charge at about 1.5 cm depth, virtually all of the salts remain in the solution, and can be flushed out by the refilling operation.
MATERIAL REQUIREMENTS OF BASIN STILLS
The materials used for this type of still should have the following characteristics:
- Materials should have a long life under exposed conditions or be inexpensive enough to be replaced upon degradation.
- They should be sturdy enough to resist wind damage and slight earth movements.
- They should be nontoxic and not emit vapors or instill an unpleasant taste to the water under elevated temperatures.
- They should be able to resist corrosion from saline water and distilled water.
- They should be of a size and weight that can be conveniently packaged, and carried by local transportation.
- They should be easy to handle in the field.
Although local materials should be used whenever possible to lower initial costs and to facilitate any necessary repairs, keep in mind that solar stills made with cheap, unsturdy materials will not last as long as those built with more costly, high-quality material. With this in mind, you must decide whether you want to build an inexpensive and thus short-lived still that needs to be replaced or repaired every few years, or build something more durable and lasting in the hope that the distilled water it produces will be cheaper in the long run. Of the low-cost stills that have been built around the world, many have been abandoned. Building a more durable still that will last 20 years or more seems to be worth the additional investment.
Choosing materials for the components in contact with the water represents a serious problem. Many plastics will give off a substance which can be tasted or smelled in the product water, for periods of anywhere from hours to years. As a general guide, if you are contemplating using any material other than glass or metal in contact with water, you may perform a useful screening test by boiling a sample of the material in a cup of good water for half an hour, then let the water cool, and smell and taste it. This is a considerably accelerated test of what happens in the still. If you can tell any difference between the test water and that you started with, the material is probably safe to use. To get some experience, try this on polyethylene tubing, PVC pipe and fiberglass resin panel.
Basic Components
A basin still consists of the following basic components: (1) a basin, (2) support structures, (3) glazing, (4) a distillate trough, and (5) insulation. The section that follows describes these components, the range of materials available for their construction, and the advantages and disadvantages of some of those materials.
The Basin. The basin contains the saline (or brackish) water that will undergo distillation. As such, it must be waterproof and dark (preferably black) so that it will better absorb the sunlight and convert it to heat. It should also have a relatively smooth surface to make it easier to clean any sediment from it.
There are two general types of basins. The first is made of a material that maintains its own shape and provides the waterproof containment by itself or with the aid of a surface material applied directly to it. The second type uses one set of materials (such as wood or brick) to define the basin's shape. Into this is placed a second material that easily conforms to the shape of the structural materials and serves as a waterproof liner. No one construction material is appropriate for all circumstances or locations. Table 1 lists the various materials and rates them according to properties desirable for this application.
Table 1. A Comparison of Various Materials Used in Solar Basin Construction
Type of Dura- Local Avail- Skill Port- Toxi- Material bility Cost ability Needed Cleaning ability city
Enameled High High Low Low High Medium Low Steel
EPDM High High Low Low High High Low Rubber
Butyl High High Low Low High High Low Rubber
Asphalt High Medium Medium Medium Medium Medium [a] Mat
Asbestos High Medium Low Medium Medium Medium High Cement
Black Medium Low Low Low Medium High Low Poly- ethylene
Roofing Medium Medium High Medium Medium Low [a] Asphalt on Concrete
Wood Low [a] [a] Medium Medium Medium Low
Formed Medium Medium Low Low High Medium Low Fiber- glass
[a] = Unknown or depends upon local conditions.
Selecting a suitable material for basin construction is the biggest problem in the solar still industry. The corrosion conditions at the water line can be so severe that basins made of metal--even those coated with anti-corrosive materials--tend to corrode. Basins made of copper, for example, are likely to be eaten out in a few years. Galvanized steel and anodized uncoated aluminum are likely to corrode in a few months. This is also true of aluminum alloys used to make boats. There are many chemical reactions that double in rate with each 10 [degrees] centigrade increase in temperature. Whereas an aluminum boat might last 20 years in sea water at 25 [degrees] C if you increase that temperature by 50 [degrees], the durability of that aluminum may well be only one or two years.
Porcelain-coated steel lasts only a few years before it is eaten out by corrosion. The special glass used for porcelain is slightly soluble in water, and inside a still it will dissolve away. The typical life of stills equipped with porcelain basins is about five years, although several have been kept operating much longer than that by repairing leaks with silicone rubber.
People have also tried to use concrete because it's inexpensive and simple to work with, but the failure rate has been high because it often develops cracks if not during the first year, then later on. Concrete and abestoscement also absorb water. The water may not run right on through, but it does soak it up. Everybody knows that satisfactory cisterns and reservoirs are built of concrete, but in a solar still the rules change. Any part of it that is exposed to outside air will permit evaporation. Since it is salt water that is being evaporated, salt crystals will form in the concrete near the surface and break it up, turning it to powder.
What about plastic? Every few years, someone decides that if we could just mold the whole still--except for the glass and glass seal--out of some plastic such as styrofoam, it would be so easy and inexpensive. But styrene foam melts at about 70 [degrees] Centigrade. Urethane foam is a little more promising, but it tends to be dimensionally unstable, and, if a still is constructed in the inclined-tray configuration, the efficiency suffers, because the non-wetted portions do not conduct heat to the wetted portions very well.
What about fiberglass? People have spent a lot of time trying to build stills from fibreglass resin formulations. Thus far, they have found the material to be unusable for any part of the still (e.g., the basin or distillate trough) that comes in contact with water, either in liquid or vapor form. Epoxy and polyester resins can impart taste and odor to the distilled water, not just for weeks, but for years. Researchers have found that this problem cannot be eliminated by covering these materials with a coat of acrylic br anything else. The odors migrate right through the coating and make the distilled water unsalable, if not undrinkable. Moreover, using fiberglass resin is not a particularly low-cost approach. Finally, a fiberglass basin or trough that is subjected to hot water for many years develops cracks. Unless researchers find a way to solve these problems, fiberglass remains an unsuitable material.
One alternative is ordinary aluminum coated with silicone rubber. The durability of basins made with this material increased into the 10- to 15-year range. For the hundreds of stills one company sold using this material, the coating was all done by hand. With production roll coating equipment, the basin's durability could probably be increased even more.
Although stainless steel has been used, success has been poor.
Support Structures. Support structures form the sides of the still as well as the basin, and support the glazing cover. As noted earlier, some materials used in forming the basin also form the still support structure while other still configurations demand separate structures, especially to hold the glazing cover.
The primary choices for support structures are wood, metal, concrete, or plastics. In most cases the choice of material is based upon local availability. Ideally, the frame for the glazing cover should be built of small-sized members so they do not shade the basin excessively.
Wooden support structures are subject to warping, cracking, rot, and termite attack. Choosing a high-quality wood, such as Cypress, and letting it age may help to alleviate these problems, but, if high heat and high humidity prevail inside and outside the still, the still will require frequent repair or replacement. The main advantage of wood is that it can be easily worked with basic hand tools.
Metal may be used for the supports but is subject to corrosion. Since metals are not subject to warping, they can aid in maintaining the integrity of the seals, although the expansion rate of a metal must be taken into account to ensure its compatibility with the glazing material and any sealants used. Use of metal for frame members is practically limited to aluminum and galvanized steel. Both will last almost indefinitely, if protected from exposure.
Silicone rubber will not adhere well to galvanized steel, but does so very well to aluminum.
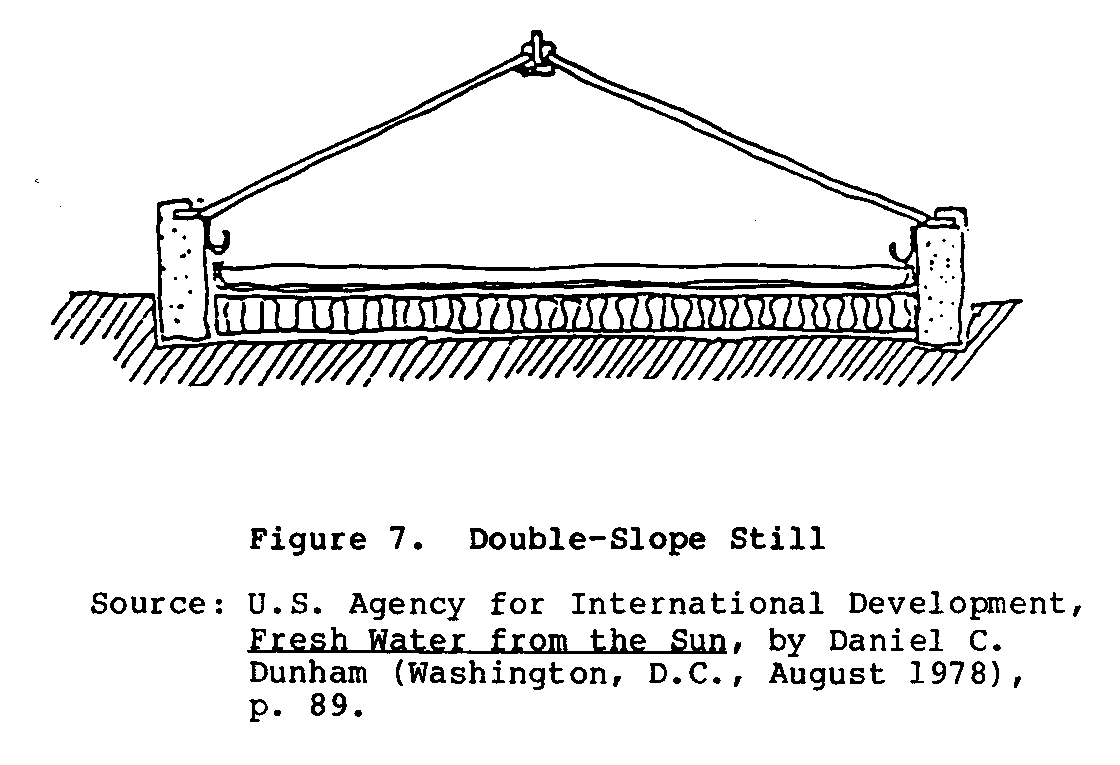
Concrete and other masonry materials may form the sides and glazing support of a still as well as the membrane. This is more readily possible in a single-slope still (Figure 6) rather than

in a double-slope still (Figure 7).
Glazing Cover. After the pan, the glazing cover is the most critical component of any solar still. It is mounted above the basin and must be able to transmit a lot of light in the visible spectrum yet keep the heat generated by that light from escaping the basin. Exposure to ultraviolet radiation requires a material that can withstand the degradation effects or that is inexpensive enough to be replaced periodically. Since it may encounter temperatures approaching 95 [degrees] celsius (200 [degrees] F), it must also be able to support its weight at those temperatures and not undergo excessive expansion, which could destroy the airtight seals. A film type cover, which must be supported by tension or air pressure, seems like a very poor choice.
Ideally, the glazing material should also be strong enough to resist high winds, rain, hail, and small earth movements, and prevent the intrusion of insects and animals. Moreover, it must be "wettable." Wettability allows the condensing vapor to form as sheets of water on the underside of a glazing cover rather than as water droplets. If the water does form as droplets, it will reduce the performance of the still for the following reasons:
- Water droplets restrict the amount of light entering the still because they act as small mirrors and reflect it back out.
- A percentage of the distilled water that forms as droplets on the underside will fall back into the basin rather than flow down the glazing cover into the collection trough. Except for temporary conditions at startup, such a loss of water should not be tolerated.
Other factors determining the suitability of glazing material include the cost of the material, its weight, life expectancy, local availability, maximum temperature tolerance, and impact resistance, as well as its ability to transmit solar energy and infrared light. Table 2 compares various glazing materials based on these factors.
Of the glazing materials listed in Table 2, tempered glass is the best choice in terms of wettablity and its capability to withstand high temperatures. It is also three to five times stronger than ordinary window glass and much safer to work with. One disadvantage of tempered glass is its high cost. While tempered low-iron glass, in one series of tests, gave 6 percent additional production, it also added about 15 percent to the cost of the still. Moreover, glass cannot be cut after it has been tempered. Nevertheless, it is a valid choice, certainly for a top-quality, appliance type product.
Table 2. A Comparison of Various Glazing Materials Used in Building Solar Stills
Type Estimated Solar Infrared Light Glazing Cost(a) Weight Life Maximum Transmittance Transmittance Impact Local Material (Dollars/[Ft.sup.2]) (Lb/[Ft.sup.2]) Expectancy Temperature (Percent) (Percent) Resistance Wettability Availability
Tempered Low-Iron 1.6 to 400 [degrees]-600 [degrees] F Glass 3.60 2.5 50+ years 204 [degrees]-316 [degrees] C 91 less than 2 Low Excellent No
Ordinary Window 400 [degrees] F Glass .95 1.23 50 years 204 [degrees] C 86 2 Low Excellent Yes
Tedlar .60 .029 5-10 years 225 [degrees] F 107 [degrees] C 90 58 Low Treatable No
Mylar ? ? ? ? ? ? Low Treatable No
Acrylic 1.50 .78 25+ years 200 [degrees] F 93 [degrees] C 89 6 Medium Treatable No
Polycarbonate 2.00 .78 10-15 years 260 [degrees] F 127 [degrees] C 86 6 High Treatable No
Cellulose Acetate 180 [degrees] F Butyrate .68 .37 10 years 82 [degrees] C 90 ? Medium ? No
Fiberglass .78 .25 8-12 years 200 [degrees] F 93 [degrees] C 72-87 2-12 Medium Treatable No
Polyethylene .03 .023 8 months 160 [degrees] F Possibly 71 [degrees] C 90 80 Low treatable ?
(a) Costs are in U.S. dollars, and were developed based on data published between 1981 and 1983. Ordinary window glass is the next best choice, except that it has an oily film when it comes from the factory, and must be cleaned carefully with detergent and/or ammonia. If you choose glass as a glazing material, double-strength thickness (i.e., one-eighth of an inch, or 32 millimeters) is satisfactory.
While some plastics are cheaper than either window glass or tempered glass, they deteriorate under high temperatures and have poor wettability. Moreover, under temperature conditions typical of solar stills, the chemicals in plastics are likely to interact with the distilled water, possibly posing a health hazard.
What about the size of the glass? Using a low slope of glass, the goal is to make it as wide from north to south as possible. It doesn't take any more labor to install a 90 centimeter piece of glass than it does to install one of 60 centimeters and you get more absorber area. Also, loss of heat through the walls will be the same whether the still is large or small. Using pieces of glass wider than 90 centimeters (3 ft.) introduces two problems: (1) the price per unit area of the glass goes up; and (2) the labor costs and the danger of handling it increase. On the basis of experience, one optimal size is about 86 centimeters (34"), a size that is commonly stocked and widely available, especially in the solar collector industry.
Distillate Trough
The distillate trough is located at the base of the tilted glazing. It serves to collect the condensed water and carry it to storage. It should be as small as possible to avoid shading the basin.
The materials used for the trough must satisfy the general material requirements outlined previously. Those most commonly used include metal, formed materials used in basin construction (with or without plastic liners), or treated materials.
Stainless steel is the material of choice, although it is expensive. Common varieties, such as 316, are acceptable. Other metals require protective coatings to prevent corrosion. Aluminum is not supposed to corrode in distilled water, but it seems preferable to rub a coating of silicone rubber over it anyway. Galvanized iron probably will not last more than a few years at most, and copper and brass should not be used because they would create a health hazard. Also, steel coated with porcelain is a poor choice because the glass will dissolve slowly and allow the steel to rust.
Basins lined with butyl rubber or EPDM can have their liners extend beyond the basin to form the trough. This method is inexpensive to implement and provides a corrosion-free channel.
No version of polyethylene is acceptable because it breaks up and emits an unpleasant odor and taste. Some people have used polyvinyl chloride (PVC) pipe, slit lengthwise. However, it is subject to significant distortion inside the still, can give off an undesirable gas, and is subject to becoming brittle when exposed to sunlight and heat. Butyl rubber should be okay, but because it is black, the distillate trough becomes an absorber and re-evaporates some of the distilled water (a minor problem).
Ancillary Components
Ancillary components include insulation, sealants, piping, valves, fixtures, pumps, and water storage facilities. In general, it is best to use locally available materials, which are easily replaceable.
Insulation
Insulation, used to retard the flow of heat from a solar still, increases the still's performance. In most cases, insulation is placed under the still basin since this is a large area susceptible to heat loss.
In stills where the depth of water in the basin is two inches or less, performance has been increased by as much as 14 percent, but this gain decreases as the depth of the water in the basin increases. Increases in performance resulting from the installation of insulation materials are also less in those locations where greater amounts of solar energy are available.
The least expensive insulation option is to build a solar still on land that has dry soil and good drainage. The use of sand helps to minimize solar heat losses, and may also serve as a heat sink, which will return heat to the basin after the sun sets and prolong distillation process.
Insulation, which adds approximately 16 percent to construction costs, may be extruded styrofoam or polyurethane (Note: polyurethane in contact with soil will absorb moisture and lose much of its insulation value.)
Sealants
Although the sealant is not a major component of a solar still, it is important for efficient operation. It is used to secure the cover to the frame (support structure), take up any difference in expansion and contraction between dissimilar materials, and keep the whole structure airtight. Ideally, a good sealant will meet all of the general material requirements cited earlier in this paper. Realistically, however, it might be necessary to use a sealant that is of lesser quality and has a shorter lifespan but that may be locally available at prices more affordable to people in developing countries. One major drawback of applying low-cost sealants to stills is the frequent labor input the stills require to keep them in serviceable condition.
Sealing a solar still is more difficult than sealing a solar water-heating panel on two counts: (1) an imperfect seal could cause a drop of rain water carrying micro-organisms to enter the still, which would contaminate the water; and (2) applying a sealant that imparts a bad taste or odor to the distilled water will make it unpalatable.
Traditional sealants that are locally available include:
- window putty (caulk and linseed oil);
- asphalt caulking compound;
- tar plastic;
- black putty.
A wide variety of other caulks sealants is also available. These include latex, acrylic latex, butyl rubber and synthetic rubbers, polyethylene, polyurethane, silicone, and urethane foam. Most of these will be more costly than traditional varieties, but they may wear longer.
Of this group of sealants, molded silicone or EPDM, clamped in place, seems to be the most promising. Silicone rubber sealant, applied from a tube, is certainly a superior choice, although people have reported a few instances of degradation and seal failure after 5 to 15 years when the seal was exposed to sunlight. Covering the sealant with a metal strip should extend its life greatly. Researchers are experimenting with an extruded silicone seal, secured by compression.
One final note: Remember a sealant that works well for windows in a building does not assure that it will work in a solar still, due to higher temperatures, presence of moisture, and the fact that the water must be palatable and unpolluted.
Piping
Piping is required to feed water into the still from the supply source and from the still to the storage reservoir. The general material requirements cited earlier hold true for this component.
While stainless steel is preferred, polybutylene is a satisfactory pipe material. Black polyethylene has held up well for at least 15 years as drain tubing. Nylon tubing breaks up if exposed to sunlight for 5 to 10 years. PVC (polyvinyl chloride) pipe is tolerable, although during the first few weeks of still operation it usually emits a gas, making the distilled water taste bad. Ordinary clear vinyl tubing is unacceptable. There is a "food grade" clear vinyl tubing that is supposed to be satisfactory for drinking water, but the sun's rays are likely to degrade it if it's used in a solar still. Companies sell drinking water and milk in high-density polyethylene bottles, and have had satisfactory results. But put the same plastic bottle filled with water in the sun, and the plastic will degrade, imparting a bad taste to the water. Few plastics can withstand heat and sunlight. Brass, galvanized steel, or copper may be used in the feed system, but not in the product system.
One final note: Although a solar still repeatedly subjected to freezing will remain unharmed, drain tubes so exposed may freeze shut unless you make them extra large. Feed tubes can easily be arranged with drain-back provision to prevent bursting.
Fittings
Fittings are connection devices that hold pipe segments together. If you put a solar still on the market with instructions to consumers that connections be made "finger tight only", people could put a wrench on a connection, loosen it, and be faced with an expensive repair problem. So, the options include having tight control of installation personnel, or doing a thorough training job, or making the equipment rugged enough to withstand ordinary plumbing practice.
A solar still is fed on a batch basis for an hour or two every day. It is necessary to admit some extra water each day, to flush out the brine. There is very little pressure available to get the water to drain, so drainage cannot proceed rapidly. To prevent flooding, it's good practice to insure that the feed rate does not exceed this maximum drainage rate. If one uses needle valves thus to restrict the flow, such valves have been found to be unstable over the years, generally tending to plug up and stop the flow. It has proven to be a satisfactory solution to this problem--when feeding from city water pressure of typically 50 p.s.i.--to use a length of small diameter copper tubing, such as 25 feet or more of 1/8 inch outside diameter, or 50 feet of 3/16 inch outside diameter tubing, to serve as a flow restrictor. It needs to have a screen ahead of it, such as an ordinary hose filter washer, with 50 mesh or finer stainless steel screen, to prevent the inlet end from plugging.
Storage Reservoir
In selecting materials for the storage reservoir, two precautions should be noted.
1) Distilled water is chemically aggressive, wanting to dissolve a little of practically anything, until it gets "satisfied," and then the rate of chemical attack is greatly slowed. What this number is, in terms of parts per million of different substances, is not well documented, but the practical consequences are that some things, such as steel, galvanized steel, copper, brass, solder, and mortar, which distilled water, resulting in damage or destruction of the tank component, and quite possibly in contamination of the water. Stainless steel type 316) is a good choice. Polypropylene laboratory tanks are okay but must not be exposed to sunlight. Butyl rubber lining of some structural framework should be okay. Galvanized steel would last for only a few years, adding some zinc and iron to the water. Concrete should serve, again with the expectation that the concrete will slowly crumble over many years' time. The tiny amount of calcium carbonate that is leached out can be used by the body in the diet. In fact, one way to prevent such chemical attack is to introduce some limestone or marble chips into the distilled water stream, or in the reservoir itself, to pick up some calcium carbonate on purpose, thus greatly slowing the attack on the tank itself.
2) Extreme precautions need to be taken to prevent entry of insects and airborne bacteria. Air must leave the reservoir every time water enters it, and must re-enter every time water is drawn off. Use a fine mesh--50 x 50 wires to the inch--or finer screen covering the vent, and turn the opening of the vent/screen assembly downward, to prevent entry of rain water. If this is ignored for even one hour, an insect can get in, and you have germ soup from then on.
Storage capacity should be adequate to contain four to five times the average daily output of the still.
Factors to Consider in Selecting Materials for Basin Still
Let us review the functions of the basin:
- It must contain water without leaking.
- It must absorb solar energy.
- It must be structurally supported to hold the water.
- It must be insulated against heat loss from the bottom and edges.
An infinite number of combinations of materials will serve those functions. The membrane that holds water, for example, may be stiff enough to support the water, but it doesn't have to be. The basin may be rigid enough to support the glass, but it doesn't have to be. In short, a component need not satisfy two functions at the same time. Indeed, it is usually better to select local material that will best do each job separately, and then put them together. But if you can find a material that does a couple of jobs well, so much the better.
In selecting materials for a solar still, there are almost always tradeoffs. You can save money on materials, but you may lose so much in productivity or durability that the "saving" is poor economy.
Summary of Materials Recommended for Basin Still Construction
Where the objective is the lowest cost of water on a 20-year life cycle cost basis, the best materials for building a basin still are:
- silicone compound coating to blacken the bottom of the basin;
- metal ribs spaced 40 centimeters (16 inches) apart to support the underside of the basin;
- about 25 to 38 millimeters of insulation between the ribs (this may be high-temperature urethane foam, or fiberglass);
- a bottom covering of lightweight galvanized steel, or aluminum sheet (note: if you plan to put the still on the ground and use an insulation that is impervious to water, no bottom sheet is needed); + metal siding, such as extruded aluminum, to support the still (note: extruded aluminum can be assembled quickly, but it is expensive; thus, you may prefer a lower cost material such as painted steel or aluminum; + a stainless steel trough; + tempered low-iron glass, or regular double-strength window glass. (If using patterned glass, put the pattern side down);
- extruded gaskets, compressed into final position; + type 316 stainless steel fittings (note: brass is not acceptable; PVC is acceptable, but poor in very hot climates);
- a mirror behind the still for higher latitudes.
Although these materials are representative of a high-cost still design, they are probably a good investment since none of the inexpensive designs has stayed on the market. However, we must also ask the question, "Expensive compared to what?" Compared to hauling purified water in bottles or tanks, solar distilled water would almost always be much less expensive. Compared to hauling vegetables by airplane to hot desert places, using a solar still to raise vegetables in a greenhouse should be less expensive.
Compared to the cost of boiling water to sterilize it, the solar still should be competitive in many situations. And although the materials used in building a high-cost still will probably always be expensive, mass production could ultimately drive down the unit cost per still.
IV. OPERATION AND MAINTENANCE OF SOLAR STILLS
OPERATING REQUIREMENTS OF BASIC STILLS
Protecting Distilled Water from Contamination
Protecting a solar still against the entry of insects and polluted rainwater is important. After your still is installed, you must:
- disinfect the interior of the still and tubing with chlorine compounds (adding a few spoonfuls of laundry bleach to a few liters of water does the job nicely); and + provide a vent(*) in the feed tube at the still, screened with fine stainless steel screen filter washer in a pipe fitting, turned downward to prevent entry of contaminated rainwater. If these precautions are not taken, flying insects, attracted by the moisture, might find their way in and die in the distillate trough.
Preventing contamination in a storage reservoir is a little more difficult, as the daily high temperature are not available to pasteurize the water. Nevertheless, with diligent attention to detail, the system can be used for decades without contamination.
Filling and Cleaning a Basin Still
Filling a basin still is a batch process (*), done once a day, at night or in the morning. With a still of this design, about 5 to 7 percent of the day's total distilled water is produced after sundown, so it is important to wait until the still is cold. Refilling it between three hours or more after sundown and up to one or two hours after sunrise will cause little, if any, loss of production.
(*) A vent allows air to enter and exit the still daily during the operation and refilling.
It is not necessary to drain the still completely. Refilling it with at least twice as much as it produces will normally dilute and flush it adequately. Three times as much would keep it a little cleaner, and could be worth doing, provided the cost of feed water is nominal. A rapid mechanical flushing is not required; a gentle trickle does the job.
Feeding Hot Water to a Basin Still
If a basin still is fed water that is hotter than the ambient air, the unit becomes a conventional distiller, except that it uses glass instead of copper as the condenser. If the hot water is virtually cost-free, as is geothermal or waste water, it can be well worth doing. If the feed water is heated by fossil fuels or by separate solar panels, the economics look doubtful, and the feed line tends to plug up with scale.
FACTORS INFLUENCING SOLAR STILL OPERATING PERFORMANCE
In this section, we discuss some important factors that influence the rate of production of distilled water. These include climatic factors, thermal loss factors, and solar still design factors.
Climate Factors
Radiation: Its Effect on Efficiency
The amount of solar radiation a solar still receives is the single most important factor affecting its performance. The greater the amount of energy received, the greater will be the quantity of water distilled. Figure 8 shows the rate of production of a basin still on the basis of specific solar inputs.
Solar stills produce less distilled water in winter than in summer, which is a problem. To some extent, the demand for drinking water also varies with the seasons, by as much as perhaps 2 to 1, summer over winter. But the annual sunlight variation affecting a still's solar distillation rate is greater than that, at least in regions well outside the tropics. In the tropics, at latitudes of less than 20 [degrees], the annual sunlight variation is probably well under 2 to 1, so it may not be a problem there. The farther away from the equator, the greater the annual sunlight variation, to perhaps 7 to 1 at 40 [degrees] latitudes. This is unacceptable, making use of a solar still difficult in winter at high latitudes.
(*) Note that there are other methods available for large distillation plants. However, because they fall outside the scope of this paper, they are not discussed here. Many approaches have been tried to solve this problem. Tilting the whole still up to more or less an equatorial mount brings the ratio down very nicely. This is called the "inclined-tray" still, and is accomplished by using many small pans in a stair-step arrangement. With this arrangement, total sunlight striking the aperture of the glass remains more constant, and the light which glances off the water of one small tray warms the bottom of the one above it, improving performance. While this is a substantial advantage, it is the only advantage of this design, and it must be weighed against the disadvantages of higher costs associated with putting many small pans vs. only one in the enclosure, and, most probably, higher installation costs due to holding the end of the pan higher off the supporting surface, and protecting it against wind loads. In latitudes perhaps 20 [degrees] on up, it seems possible that the inclined-tray will find a place in the market.
Using an inclined-tray still is only one solution to the problem of annual variation in higher latitudes. Some other steps that can be taken include:
- buying an extra large still that produces enough distilled water in winter, resulting in a likelihood that you will have more water than you need in summer;
- using less water in winter and/or using some tap water;
- buying supplemental water in winter; or
- saving some of the excess distilled water made in summer or fall for use in winter;
- installing a mirror behind the basin to reflect additional sunlight back into the still in winter. To reflect back as much light as possible, use a reflective surface of about one-third to one-half of the aperture of the glass cover, tilted forward 10 [degrees] from the vertical, mounted at the rear edge of the still. In latitudes between 30 [degrees] and 40 [degrees], this gives from 75 to 100 percent more yield in mid-winter.
Condensing-Surface Temperature. Much work has been done to try to obtain lower condensing temperatures, thereby increasing the temperature difference between the heated feed water and the condensing surface. This approach undoubtedly derives from 100 years of steam power engineering, in which it is most important to get the steam temperature high and the condensing temperature low to gain efficiency. But this principle does not hold true for a solar still. Steam for power is pure steam, whereas the contents of a solar still are both air and water vapor. It has been demonstrated repeatedly that the higher the operating temperature of the still--insolation being equal--the higher the efficiency. For each 6 [degrees] celsius (10 [degrees] F) increase in ambient temperature, the production of a still increases by 7 to 8 percent. The practical effect of this is that a still operating in a hot desert climate will produce typically as much as one-third more water than the same unit in a cooler climate.
(By the same token, cooling the glazing cover of a solar still by spraying water on it or blowing air over it does not help the still produce more distillate. In an experiment at the University of California in the United States, two identical stills were built. The glazing cover of the first still was fan-cooled; the cover of the second still was not. Of the two stills, the cooled unit produced significantly less distillate. Consequently, it's better to put the still in a protected area rather than a windy area.)
Thermal Loss Factors
Production is also associated with the thermal efficiency of the still itself. This efficiency may range from 30 to 60 percent, depending on still construction, ambient temperatures, wind velocity, and solar energy availability. Thermal losses for a typical still vary by season, as shown in Table 5.
Table 5. Distribution of Incoming Solar Radiation in the Distillation Process
December May Thermal Loss Factors (Percent) (Percent)
Reflection by Glass 11.8 11.8
Absorption by Glass 4.1 4.4
Radiative Loss from Water 36.0 16.9
Internal Air Circulation 13.6 8.4
Ground and Edge Loss 2.1 3.5
Re-Evaporation and Shading 7.9 14.5
[Remainder of Energy Used to Distill Water] 24.5 40.5
Direct Use of the Sun's Energy, Daniels, Farrington, 1964, Ballantine Books, page 124.
Solar Still Design Factors
Slope of the Transparent Cover
The angle at which the transparent cover is set influences the amount of solar radiation entering a solar still. When sunlight strikes glass straight on, at 90 [degrees] to the surface, about 90 percent of the light passes through. Tip the glass a little, so that it strikes at a "grazing angle" of 80 [degrees], and only a few percent is lost. But tilt it a few more tens of [degrees], and the curve goes over the hill, dropping off to practically zero at 20 [degrees] grazing angle, where virtually no direct light gets through. In a greenhouse-type still, for a large part of the year the half of the glass that is facing away from the equator is receiving sunlight at very low grazing angles. It is actually shadowing the back one-third of the still. It is more efficient to make that half of the glass facing the equator as long as possible, and put a more or less reflective back wall to the rear. This was one of the significant steps that has increased the efficiency of basin stills from 31 to about 43 percent, using a single slope of glass. And it costs less to build.
The slope of the glass cover does not affect the rate at which the distillate runs down its inner surface to the collection trough. A common misconception was that the glass cover must be tilted to get the water to run off. This may have arisen from the fact that ordinary window glass, as it comes from the factory, has a minute oily film on it. But if the glass is clean, the water itself will form filmwise condensation on it, and will be able to run off at a slope as little as 1 [degrees].
There are three reasons why it is best to use as low a slope as possible: (1) the higher the slope, the more glass and supporting materials are needed to cover a given area of the basin; (2) the higher slope increases the volume and weight [of the still] and therefore shipping costs; and (3) setting the glass at a high slope increases the volume of air inside the still, which lowers the efficiency of the system. A glass cover that is no more than 5 to 7 centimeters from the water surface will allow the still to operate efficiently. Conversely, as glass-to-water distance increases, heat loss due to convection becomes greater, causing the still's efficiency to drop.
Some important stills have been built following the low-slope design concept for the glass cover, yet using a short, steeply sloping piece of glass at the rear. This requires either providing an extra collection trough at the rear, or else making the successive troughs touching heel and toe, so that it is exceedingly difficult to get out in the middle of the array to service anything. It also increases the condensing surface relative to the absorber, which reduces operating temperatures in the still, and is clearly disadvantageous. A reflective and insulated back may be preferable to glass.
Some years ago at the University of California, researchers built an experimental multiple tray tilted still with an average glass-to-water distance of about 30 millimeters, showing an efficiency of 62 percent, one of the highest ever recorded. The loss of efficiency is greatest the first centimeter, rather less the second cm, and so on, tailing off to smaller rates of loss per cm distance as far as the test was carried. This is one of the principle reasons a high slope of glass is to be avoided.
In sum, it is clear that a solar still should be built in a way that will get the water as hot as possible, and keep it as close to the glass as possible. This is achieved by keeping the glass cover at a minimum distance from the water surface, which in practical terms, falls between 5 and 7 cm., and by minimizing the depth of water in the pan, to about 1.5 cm.
Wicks and Related Techniques
Researchers have tried to improve the efficiency of a solar still by enhancing its surface evaporation area using wicks. In a side-by-side test of two identical stills at the University of California, using a floating black synthetic fabric in one still and nothing in the other, the difference in production between the stills was indistinguishable, though similar tests have reported some improvement. It seems exceedingly difficult to find a wick material that will last for 20 years in hot saline water, and that will not get crusted up with salts over a period of time. As for putting dye in the water, studies suggest that the slight improvement in performance does not justify the increased cost and maintenance and operating problems associated with this technique.
Putting dark-colored rocks in the feedwater to store heat for use after nightfall has been reported by Zaki and his associates to improve performance by 40 percent, but he does not give the reference point from which this is measured. If he was comparing one still containing 4 cm. of water with another same water depth but containing black stones, the productivity would increase somewhat due to the decrease in thermal mass and resulting increase in operating temperature. Reducing the initial water depth might have accomplished the same result. For this reason, placing dark-colored rocks in the feedwater does not appear to be a promising technique for improvements in solar still performance.
MAINTENANCE REQUIREMENTS OF BASIN STILLS
Ways of Handling the Buildup of Mineral Deposits
It is inevitable that some minerals are deposited on the bottom of the basin. In most situations, including sea water and city tap water, the amount deposited is so small that it creates no problem for decades. One still in particular has been operated for 20 years without ever having been opened or cleaned. As long as there is not an excessive buildup of deposits, indicated by formation of a dried-out island in the afternoon, they create no problem. Such mineral deposits become the normal absorber. An accumulation of these deposits changes the interior surface of a basin from its original black color to a dark earth brown, reflecting some sunlight, causing a 10 percent drop in still production. To offset this reduction, simply make the still 10 percent larger than it would need to be if it were cleaned out periodically.
Some desert waters high in alkalis will deposit a whitish gray scale on the bottom and sides of a basin. In fact, almost any feed water will do so, especially if the basin is allowed to dry out. In some cases, the alkaline water may form a crust of scale which is held on the water's surface by air bubbles that are discharged when the feed water is heated. Light-colored deposits such as these may reduce production of the still by 50 percent or more. Those that settle to the bottom of the basin can be easily coated black by mixing one tablespoon of black iron oxide concrete coloring powder with about 10 or 15 liters of water and adding the solution to the still by means of a funnel connected to the feed water pipe. This blackening agent is inert, and imparts no bad taste or odor to the distilled water. After the solution reaches the basin through the feed water pipe, it settles on the bottom of the basin and restores it to its original black color. Some owners do this each fall, when production begins to drop. Cost is only pennies per application.
Deposits that float on the surface of the water in a basin are a tougher problem and one that requires more research. An Australian solar still expert suggests agitating the contents of the still by recirculating, or stirring, the water in the pan for one hour each night, to minimize the buildup of floating deposits. Adding a pint or two of hydrochloric (swimming pool) acid to the still whenever the bottom becomes grayish-white--every year or two, maybe oftener in some cases--is a satisfactory way of removing practically all of the scale.
Accumulation of Dust on the Glazing Cover: What to Do
In the vast majority of stills, dust accumulates on the glass cover. But it does not keep building up; it's held more or less constant by the action of rain and dew. This "normal" accumulation causes production to drop from 5 to 15 percent. To offset this, simply make your still 10 percent larger than it would need to be if kept clean. However, if the still is in an unusually dusty area, or if it is large enough that a caretaker is available at modest cost, cleaning the glazing cover is justified. Ten percent of 10,000 liters per day may be enough to justify cleaning the cover once a month in the dry season.
Repair and Replacement of Basin Still Components
As with all devices, the components of a basin still may need to be repaired or replaced from time to time. The frequency depends on the type of material used to construct the still. One built with premium materials will require almost no maintenance, but will entail a higher capital cost because many of the materials must be imported materials. Use of cheaper materials subject to degradation will almost certainly lower the initial cost, but will increase the amount of maintenance. Even so, if the long-term cost of maintenance and the lower initial cost are less than the higher initial cost for premium materials, this may present a better option, especially if cost of capital is high. This is called "life cycle cost analysis," and it is strongly recommended.
SKILLS REQUIRED TO BUILD, OPERATE, AND MAINTAIN A BASIC STILL
Craftmanship and attention to detail in construction are important for an efficient, cost-effective still.
In addition, supervisory personnel must be on hand who know how to size stills to meet a community's water supply needs; who know how to orient stills; who are familiar with required construction techniques; and who have the ability to train others in the construction, operation and maintenance of stills.
Finally, it is important to ask local workers to participate in the planning and construction phases of a solar still project to get the indigenous population to accept the technology. A sense of pride in the building of the project may well mean the difference between long-term success or failure of the project.
COST/ECONOMICS
The cost and economics of solar stills depend on many variables, including:
- cost of water produced or obtained by competing technologies;
- water requirements;
- availability of sunlight;
- cost of locally-available materials;
- cost of local labor;
- cost of imported materials; and
- loan availability and interest rates.
Table 6 shows the variation in costs for stills built in the 1970s in the Philippines, India, Pakistan, and Niger. Note that stills built in Niger in 1977 cost twice as much as those built in the Philippines in the same year, reflecting the wide variation in local cost.
Table 6. Variation in Costs for Stills Built in the 1970s
Location Year Built (Dollars/Square Foot)
Philippines 1977 $3.56
India 1975 1.39
Pakistan 1973 1.37
Niger 1977 6.30
(Costs today are undoubtedly higher.)
WHY BUY A STILL?--It saves money.
A solar still must operate with extremely low costs for maintenance arid operation. Over a long period according to a study by George Lof, it is valid to assume that 85 percent of the cost of water from the still will be chargeable to the costs of buying it; the remainder to operation and maintenance.
It is easy to calculate the return on investment in a solar still. Say you have one that produces a daily amount of water that would cost you $1 to buy in bottles: then that still returns you $365 per year. If the still had cost you $365, then it paid for itself in one year; if five times that much, then five years, etc.--not counting interest. Cost of feeding water into it is pretty small, but will increase the payout period a little also. In the United States, the payout period tends to run between two and five years, depending on the still's size and features.
SPECIAL DESIGN VARIATIONS
The majority of information presented thus far has centered on the basin-type solar still because it is the easiest to construct and may use a wide range of materials, making it adaptable to different locales. But variations of the basin still are possible, such as the double-slope and single-slope stills depicted earlier in this paper. In addition to these options, there are other ways to design the still to increase its efficiency or potential to produce potable water. Some of these are discussed below.
Basin Stills Equipped with Reflectors
Some stills have been equipped with reflective materials which have the potential to increase the amount of sunlight falling on the still without having to increase the area of the still. At latitudes in the thirties, performance increases in winter of 100% have been achieved with a mirror of less than 1/2 the area of the glass. In the tropics, of course, this function is not required. A second question arises about using mirrors to enhance production year round. This becomes a focusing collector, which introduces substantial additional costs and problems. If the mirror assembly is cheaper than the pan assembly, then it deserves to be looked at further, but it is not attractive at this time. Tentatively, reflective aluminum sheet has the most advantages.
Basin Stills Equipped with Insulated Glazing Covers
Another innovation is the use of an insulated glazing cover to be put over the glazing at night or during extremely cold weather. This cuts heat losses, allowing distillation to continue longer, and retains heat overnight, causing production to start earlier the next day. Cost-benefit analysis of this approach has not been made. V. COMPARING THE ALTERNATIVES
For a couple of gallons of purified water a day, there is no method that can compete with solar distillation. For a couple of million gallons a day--AS LONG AS WE ARE WILLING TO BURN UP OUR INHERITANCE OF FOSSIL CHEMICAL BUILDING BLOCKS JUST TO EVAPORATE WATER--boiling distillation is the cheapest way to purify sea water.
In sum, solar stills have:
- high initial costs;
- the potential to use local materials;
- the potential to use local labor for construction and maintenance;
- low maintenance costs (ideally);
- no energy costs (not subject to fuel supply interruptions);
- few environmental penalties; and
- in residential sizes, no subsequent costs for delivering water to the end user.
Most competing technologies are:
- low in initial costs;
- dependent on economy of scale;
- high in operating and maintenance-costs;
- high in energy input costs;
- low in local job creation potential;
- vulnerable to changes in energy supply and costs; and
<Figure 9>


<Figure 10>
<Figure 11>
VI. CHOOSING THE TECHNOLOGY RIGHT FOR YOU
FACTORS TO CONSIDER
Solar energy is an excellent choice for water distillation in those areas of the Third World that meet the following conditions:
- expensive fresh water source (US) $1 or more per 1,000 gallons);
- adequate solar energy; and
- available low-quality water for distillation.
Other conditions suitability for solar stills are:
- competing technologies that require expensive conventional wood, or petroleum fuels;
- isolated communities that may not have access to clean water supplies;
- limited technical manpower for operation and maintenance of equipment;
- areas lacking a water distribution system; and
- the availability of low-cost construction workers.
The greater the number of these conditions present, the more solar stills are likely to be a viable alternative. If the cost of the water produced by a still over its useful life is less than by alternate methods, it is economical to pursue.
Other factors to consider are the availability and cost of capital, as well as the local tax structure, which may allow tax credits and depreciation allowances as a means to recover a portion of the cost. This has proved to be a major incentive in the United States.
Finally, the acceptance of solar distillation will depend greatly on how well one understands and handles the many social issues and cultural constraints that can hamper the introduction of new technologies. Some of the more important issues that may affect the acceptance of solar distillation are outlined below.
- Stills built for village use require community cooperation that may be foreign to some cultural groups. If the distilled water is incorrectly distributed, causing a family unit not to receive its fair share of water, this could become a source of conflict. For this reason, a family-sized solar still unit, which a household has complete control over, may be more practical than a unit that serves an entire village.
- Potential users who think they will find distilled water tasteless or not in keeping with what they are accustomed to may become disappointed and possibly abandon altogether the thought of drinking the water. The problem of taste must be dealt with early on so as not to give people a reason to respond negatively to the technology as a whole.
- In some societies, conflicts may arise over whether it is the responsibility of the man or the woman of the household to operate the solar still. Not dealing with this issue early on could result in the household's total rejection of the technology.
- If solar distillation is perceived to be a threat to a community's traditional lifestyle, the community may reject the technology. Such concerns can be headed off if the technology is designed appropriately from the start and introduced at the proper time. Moreover, a community is more likely to accept the technology if it recognizes the importance of clean water and considers it a priority to the degree that it is willing to change certain aspects of its lifestyle.
MARKET POTENTIAL
Three potential markets exist for solar stills. First, a solar still can be economically attractive almost any place in the world where water is hauled and where a source of water is available to feed the still.
Second, many people who boil their water to kill germs could use a solar still for the same purpose. It will take more work to demonstrate this function adequately, but early tests have made it seem highly promising.
A third market is in arid regions, whose untapped water resources may be sufficient to economically provide a population with potable water.
CONCLUSION
Worldwide experience in researching and marketing solar stills over three decades has provided an ample foundation for a solar still industry. No inherent technical or economic barriers have been identified. A solar still is suited to village [manufacturing] techniques and to mass production. Around the world, concerns over water quality are increasing, and in special situations a solar still can provide a water supply more economically than any other method. Commercial activities are picking up after a lull during the late 1970s. It is now possible to predict a rapid increase in the manufacture and marketing of solar stills.
SUPPLIERS AND MANUFACTURERS OF SOLAR STILLS
Lodestone Engineering P.O. Box 981 Laguna Beach, California 92652-0981 USA
SOLEFIL Tour Roussel-Nobel CEDEX No. 3 F. 92080 Paris La Defense FRANCE
Cornell Energy, Inc. 4175 South Fremont Tucson, Arizona 85714 USA BIBLIOGRAPHY
Cooper, P.I., "Solar Distillation--State of the Art and Future Prospects." Solar Energy and the Arab World (1983): 311-30.
Daniels, Farrington. Direct Use of the Sun's Energy. New York, New York: Ballantine Books, 1975.
El-Rafaie, M.E.; El-Riedy, M.K.; and El-Wady, M.A. "Incorporation of Fin Effect in Predicting the Performance of Cascaded Solar Stills." Solar Energy and the Arab World (1983): 336-40.
Goetchew, Martin. "Shedding Light on Solar Collector Glazing." Materials Engineering 90 (September 1979): 55-58.
Langa, Fred; Flower, Bob; and Sellers, Dave. "Solar Glazzings: A Product Review." New Shelter (January 1982): 58-69.
Leckie, Jim; Master, Gil; Whitehouse, Harry; and Young, Lily. More Other Homes and Garbage. San Francisco, California: Sierra Club Books, 1981.
Mohamed, M.A. "Solar Distillation Using Appropriate Technology." Solar Energy and the Arab World (1983): 341-45.
Talbert, S.G.; Eibling, J.A.; and Lof, George. Manual on Solar Distillation of Saline Water. Springfield, Virginia: National Technical Information Service, April 1970.
Dunham, Daniel C. Fresh Water From the Sun. Washington, D.C.: U.S. Agency for Internation Development, August 1978.
Zaki, G.M.; El-Dali, T.; and El-Shafiey, M. "Improved Performance of Solar Stills." Solar Energy and the Arab World (1983): 331-35.
McCracken, Horace: Only a small amount of McCracken's work has been published, but the data are available. Inquiries will be welcomed:
McCracken Solar Co. P.O. Box 1008 Alturas, California 96101 USA